Our Focus:
The goal of our research is to improve accessibility and quality of donated organs by developing a more efficient ex vivo organ support system.
Synthetic blood cells have the potential to play a role in ex-vivo organ support by increasing its viability. There are thousands of patients awaiting organ transplants, and people die everyday waiting on this list. Many attempts have been made to create synthetic blood cells that can change these statistics. There are currently no viable products available that have been approved by the Food and Drug Administration, but one such attempt, ErythroMer, is the subject of this research that focuses on testing and defining the desired limits of the cell to be used in ex vivo kidney support.
Research Plan
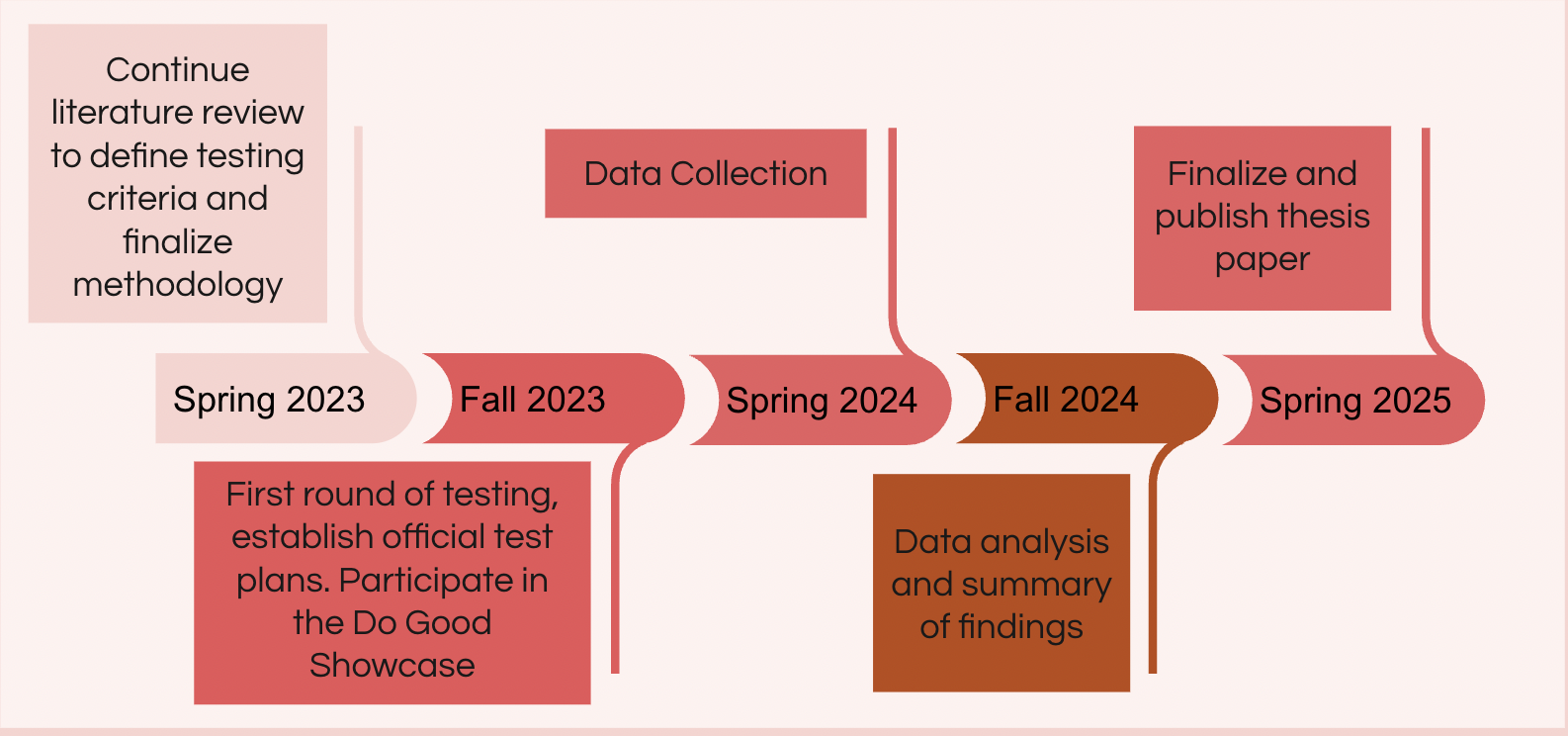
Our research is initially focused on testing the failure point of ErythroMer particles by subjecting samples to varying shear stress and measuring the free hemoglobin using a rheometer. This allows us to identify the strength of the ErythroMer particles and how long they can be subjected to stress before being unable to function properly.
After establishing a baseline failure rate for the ErythroMer particles, the next steps will be to place them under constant shear stress equal to what the pump system exerts for extended periods of time, calculating the failure rate after each experiment. Taking these data points, we will extrapolate the time the tested formulation of ErythroMer could function within the pump and how that relates to our goal. Afterwards, it will be necessary to mathematically determine the specific requirements of kidney perfusion in order to later quantify the effectiveness of the perfusate. The particle tolerance, or stability, of ErythroMer will be defined in the following ways: by the value of the absolute maximum shear stress the particle can withstand before rupturing and releasing hemoglobin into the perfusing liquid, and the amount of shear stress that the ErythroMer particle can sustainably withstand over a period of four days.
Finally, we will alter the main factors that may lead to failure in this system. This may include the actual structure of ErythroMer, the temperature under which it is most effective, and the buffer solution it is in. For instance, the proportion of Pluronic F68 could be increased to further protect the ErythroMer particles from excess drag. We may also need to modify the particles themselves: ErythroMer derives its flexibility from polyethylene glycol, and its rigidity from its lipid bilayer. Increasing these aspects may lead to better particle durability, at the cost 0f oxygen efficiency.
Our goal will be to supply a kidney with the necessary oxygen it needs to extend its viability, while maintaining a failure rate below 20%.
After establishing a baseline failure rate for the ErythroMer particles, the next steps will be to place them under constant shear stress equal to what the pump system exerts for extended periods of time, calculating the failure rate after each experiment. Taking these data points, we will extrapolate the time the tested formulation of ErythroMer could function within the pump and how that relates to our goal. Afterwards, it will be necessary to mathematically determine the specific requirements of kidney perfusion in order to later quantify the effectiveness of the perfusate. The particle tolerance, or stability, of ErythroMer will be defined in the following ways: by the value of the absolute maximum shear stress the particle can withstand before rupturing and releasing hemoglobin into the perfusing liquid, and the amount of shear stress that the ErythroMer particle can sustainably withstand over a period of four days.
Finally, we will alter the main factors that may lead to failure in this system. This may include the actual structure of ErythroMer, the temperature under which it is most effective, and the buffer solution it is in. For instance, the proportion of Pluronic F68 could be increased to further protect the ErythroMer particles from excess drag. We may also need to modify the particles themselves: ErythroMer derives its flexibility from polyethylene glycol, and its rigidity from its lipid bilayer. Increasing these aspects may lead to better particle durability, at the cost 0f oxygen efficiency.
Our goal will be to supply a kidney with the necessary oxygen it needs to extend its viability, while maintaining a failure rate below 20%.
Who is Team HEM(O-)?

Top row (Left to right):
Jackson Lang-Web Liaison
Claire Kettula-Team Liaison
Kylie Ly-Team Liaison
Bottom row (left to right):
Talya Simcox-Financial Liaison
Emily Passaro-Mentor Liaison
Joel Bannerman-Financial Liaison
Matthew Somerville-Web Liaison
Not Pictured:
Dr. Doctor-Team Mentor
Leah DiCiesare-Team Librarian
Stephen Rogers-Laboratory Advisor
Jackson Lang-Web Liaison
Claire Kettula-Team Liaison
Kylie Ly-Team Liaison
Bottom row (left to right):
Talya Simcox-Financial Liaison
Emily Passaro-Mentor Liaison
Joel Bannerman-Financial Liaison
Matthew Somerville-Web Liaison
Not Pictured:
Dr. Doctor-Team Mentor
Leah DiCiesare-Team Librarian
Stephen Rogers-Laboratory Advisor